POSTS
Lac sliding in E. Coli continued: Deeper into the mechanism
We have previously shown that the lac repressor slides along the DNA to speed up the search for its specific binding site. By combining micro- and macroscopic computational models, we have now been able to investigate the details of the sliding mechanism on the atomic scale.
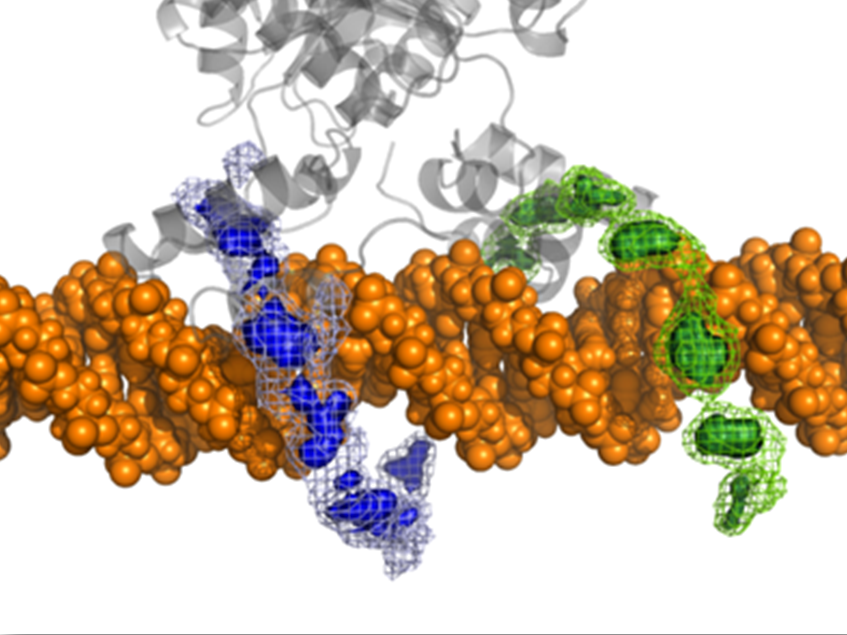
The lac dimer follows the major groove of the DNA helix, causing it to slide in a spiral motion. It remains close to the DNA for about 8bp before making a microscopic dissociation and stays on the same DNA fragment for an average 48 ms. This corresponds to an in vitro sliding length of 240 bp which correlates well with in vitro measurements. The study presents an unique combination of theoretic tools and the results satisfyingly connect macro-/mesoscopic events at the nanometer level, which enables a deeper interpretation of experimental observations.
Read more about the study in PNAS.