POSTS
It's not that simple!
One by one the mysteries of gene regulation are subject to scrutiny, and our models, the way we understand gene regulation, are put to the test. We know that lacI, the repressor of the lactose digestion machinery in the cell, searches for its binding site using a combination of 3D diffusion and sliding on the DNA, we know that it slides around 45 bases before detaching and we know that it takes less than a minute for a lacI repressor to find and bind the operator, although most of the time, the TFs slide over the binding site without binding. However, to test the prevailing model for TF mediated gene regulation, which assumes that the level of repression is determined by the equilibrium binding by the repressor to its operator, we also need to know how long the TF stays bound to the DNA before it dissociates macroscopically, e.i. leaves the immediate vicinity of the regulatory site.
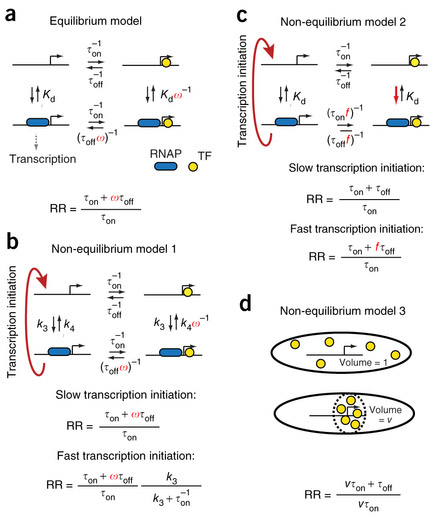
Using a new technique based on a single molecule chase assay we can answer this question - lacI stays bound to its specific operator for on average 5 min. So far so good, with an association time of 30 s and a repression ratio of 10 (as measured by an enzymatic reporter) the model is in no immediate danger. However, if the native operator is replaced by a stronger one, this results in a slower dissociation while the association rate stays approximately the same. Conclusion; our findings do not support the simple equilibrium model and this discrepancy has to be considered when predicting gene activity from TF binding strengths. For more details, visit Nature genetics