POSTS
Light in the dark
The progress in the field of live cell single molecule fluorescence microscopy that has taken place over the last couple of decades has been spectacular. However, good protocols for labeling the molecule of interest with a small, bright and photo stable fluorescent dye is limiting progress in the field, and the labeling step is where most live cell experiments fail.
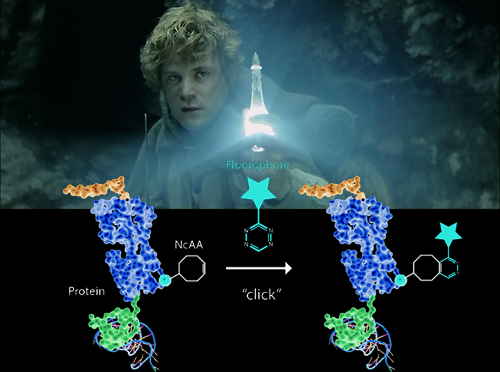
We have now explored ways to use of noncannonical amino acids to introduce handles where small organic fluorophores can be bound specifically to the protein of interest. The noncannonical amino acid is encoded via the pyrrolysyl-tRNA/pyrrolysyl-RNA synthetase pair at artificially introduced TAG codons in E. coli strains that have been recoded to lack endogenous TAG codons as well as the TAG-specific release factor RF1. The amino acids contain bioorthogonal groups that can be clicked to externally supplied dyes, thus enabling protein-specific labeling in live cells with small bright fluorophores.
The incorporation of the noncannonical amino acid in the protein of interest works well. The overall labeling scheme works well works well for proteins that spend some time on the surface of the cell and thus can be clicked to the dye extracellularly. As a model protein for the NcAA-based small fluorophore labeling we use the outer membrane porin OmpC, one of the most abundant outer membrane proteins in E.coli, which is involved in the control of cellular osmolarity and the uptake of nutrients and antibiotics. The osmoporin OmpC can be labeled sufficiently specific to enable single molecule particle tracking.
In order to label proteins in the cytoplasm however, the dye has to cross the cell membrane, which proved to be surprisingly difficult. Hydrophobic dyes can cross the membrane relatively easy, but they also interact nonspecifically with various elements in the cell and thus cause high background levels. Hydrophilic dyes are less sticky but must be forced across the membrane, a process that has severe consequences on cell viability.
Read the complete article in ACS Synthetic Biology